Four Fold Serial Dilutions4/4/2023  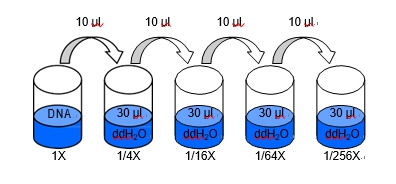
Suppose an analytical procedure states that a standard solution of 1 mg per liter is required by dilution of a 50 mg/L stock solution. In ISO, accuracy combines the concepts of unbiasedness (termed trueness) and precision (3). Accuracy in the context of this column is the International Organization for Standardization (ISO) definition. Further, it will be assumed that standard Grade A glassware are being used (2). Only the uncertainties in the volumetric glassware, however, will be considered to answer the question: “Is my dilution accurate enough?” It will be assumed that all the other factors (e.g., operator variability when pipetting or making up to the mark, temperature, and solvent effects) are constant. This column will review the preparation of standard solutions by simple and serial dilution using standard pipettes and flasks. This column looks at the effects of choice of dilution schemes on the accuracy of the prepared reference standard.Ī previous column looked at the basics of measurement uncertainty and related it to the direct preparation of a standard solution were discussed (1). Usually a stock solution is prepared followed by a dilution or series of dilutions to arrive at a working concentration of the reference standard. It is common practice to prepare such solutions volumetrically using standard glassware. Most analytical procedures rely on the accurate preparation of reference standard solutions. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
